what is double replacement in chemistry
The following is the initial neutralization reaction. The primary difference between a single and double replacement reaction is that in a single replacement reaction a free element is exchanged for another to yield a new compound and a new element.
Double Replacement Reactions Definition Examples Expii
A double replacement equation has two reactants each composed of two species and they exchange partners to form either a.
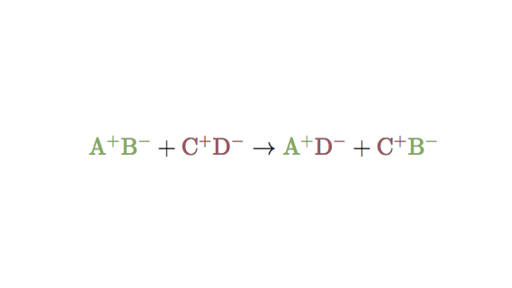
. Stay tuned with BYJUS to learn more about other concepts such as displacement reactions. Solubility rules are used to predict whether some double -. A double displacement reaction is also called a double replacement reaction salt metathesis reaction or double decomposition.
Combining vinegar and baking soda to create homemade volcano. CuCl 2 aq 2 AgNO 3 aq CuNO 3 2 aq 2 AgCls. Double replacement reactions also result in the formation of a solid product which is called a precipitate.
Chemistry Middle School answered What type of reaction is displayed below. A double replacement reaction is a chemical reaction where two reactant ionic compounds exchange ions to form two new product compounds with the same ionsDouble replacement reactions take the. Single replacement 2 See answers Advertisement Advertisement thescarletspider101 thescarletspider101 Answer.
The truefalse questions in this worksheet will help students review the process of a double-replacement reaction. Many double displacement reactions occur between ionic compounds that are dissolved in water. The reaction occurs most often between ionic compounds although technically the bonds formed between the chemical species may be either ionic or covalent in nature.
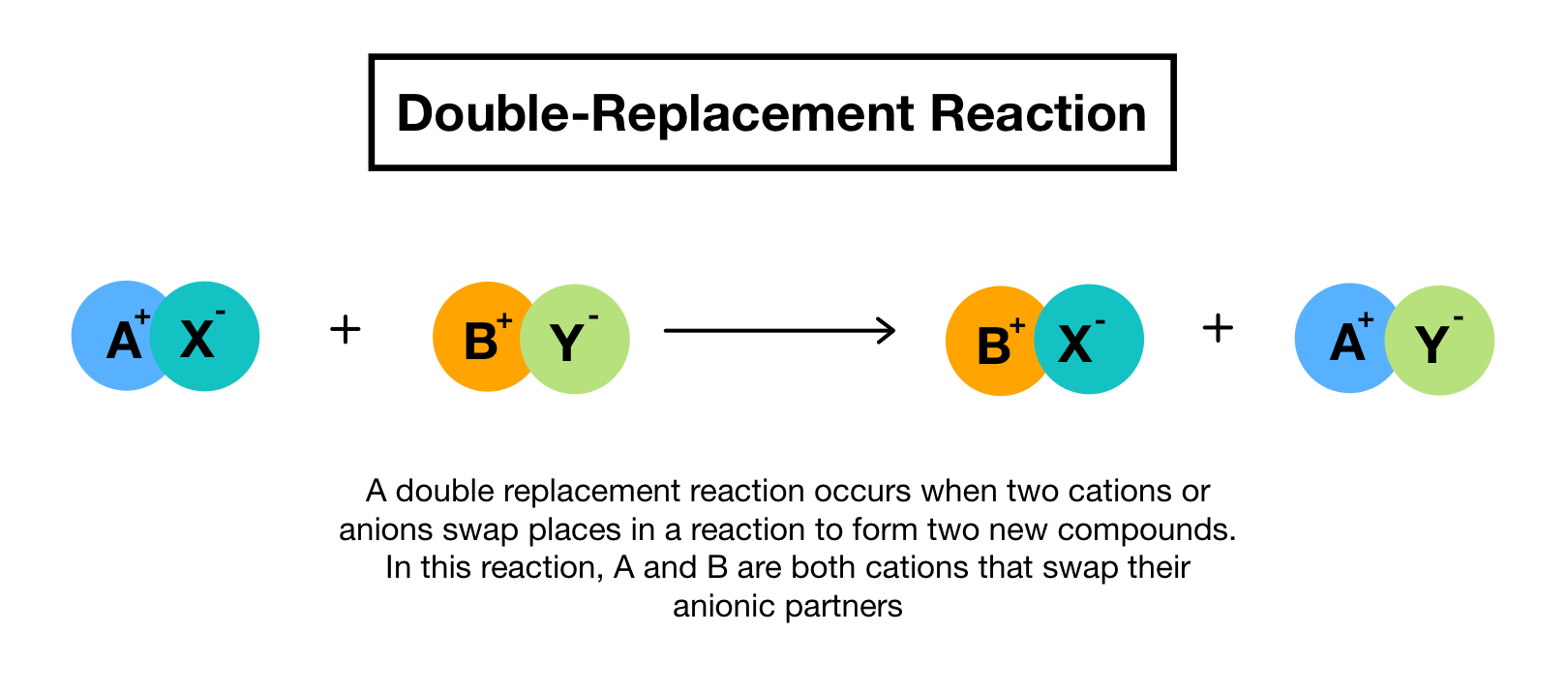
In this printable students also use an equation to answer questions about elements and chemical reactions. You can think of the reaction as swapping the cations or the anions but not swapping both since you would end up with the same substances you started with. Double replacement reaction occurs when the cations and anions of two ionic compounds are exchanged.
Two compounds react to form two new compounds. A double replacement. This is simply based on the solubility chart of inorganic compounds.
A double replacement reaction will occur if a formation of a precipitate gas or water takes place. In a double replacement reaction the components of two compounds are exchanged or displaced to form two new compounds. Double replacement reactions are also called double replacement reactions double displacement reactions or metathesis reactions.
Select two compounds above and this calculator will predict whether or not the reaction will occur in water. The following is a real life example of double replacement reactions. In double replacement reactions the elements get replaced in both the reacting compounds.
A type of reaction in which the ions of two compounds exchange places in an aqueous solution to form two new compounds Describe the nature of the reactants and products in a single replacement reaction. The figure below clearly illustrates how this swap takes place. A double replacement reaction is a type of chemical reaction that occurs when two reactants exchange cations or anions to yield two new products.
The way I think of it since were dealing with ionic compounds is that when I write out a reaction I. The solvent for a double replacement reaction is usually water and the reactants and products are usually ionic compoundsbut they can also be acids or bases. What is a double replacement displacement reaction.
Double replacement sometimes referred to as double displacement reactions are when parts of ionic compounds are switched to form two new ionic compounds. In a single replacement reaction one of the reactants is more reactive than the other which results in the formation of a product that is more stable. In double replacement reactions the positive ions exchange negative ion partners.
A characteristic of a double-replacement equation is that there are two compounds as reactants and two different compounds as products. What is a double replacement. AgNO3 KI KNO3 AgI A.
A precipitation reaction is a double - replacement reaction in which one product is a solid precipitate. Double Replacement Aqueous sodium hydroxide and hydrochloric acid Page 3 of 5 Chemistry 19 - Santa Monica College Double Replacement followed by Decomposition The product of the double displacement spontaneously decomposes into HO and CO Solid sodium bicarbonate and hydrochloric acid 6 Double Replacement Aqueous ironIII chloride and aqueous ammonium. Those reactions in which two compounds react by an exchange of ions to form two new compounds are called double displacement reactions.
A double-replacement reaction exchanges the cations or the anions of two ionic compounds. What Is a Double-Replacement Reaction. A double displacement reaction also known as a double replacement reaction or metathesis is a type of chemical reaction where two compounds react and the positive ions cation and the negative.
A double-replacement reaction occurs when parts of two ionic compounds are exchanged making two new compounds.
/GettyImages-1154749611-crop-86d1624d100d46e681f311f74fe900a2.jpg)






























